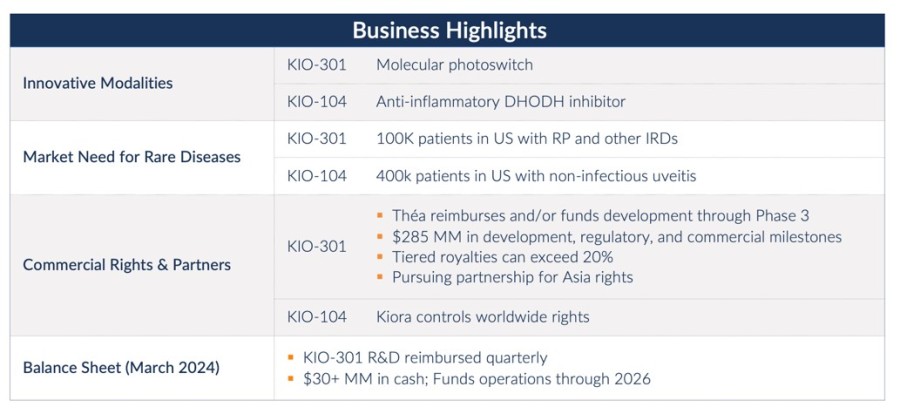
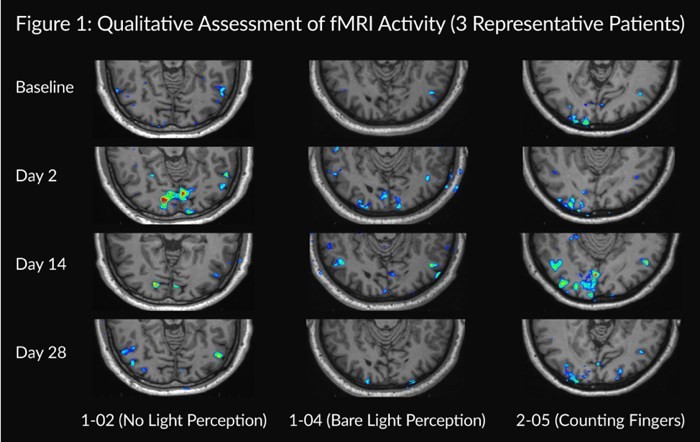
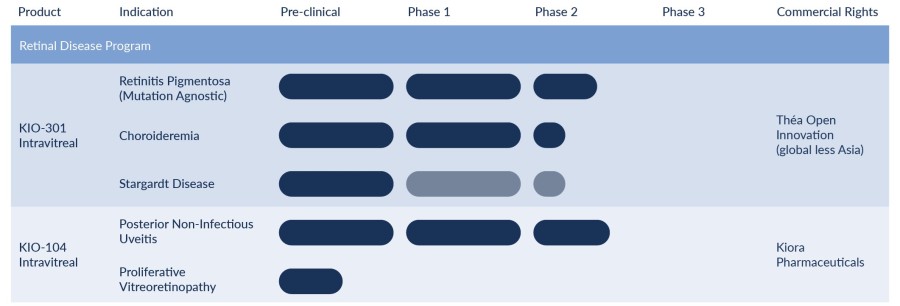
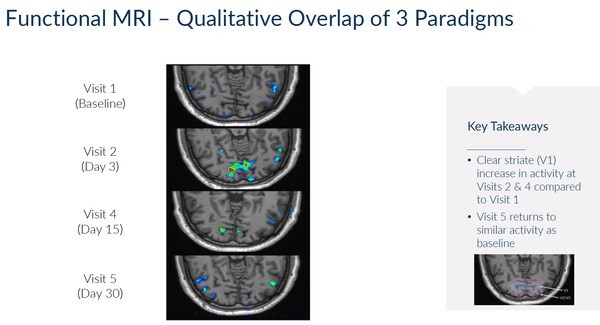
Encinitas, California--(Newsfile Corp. - November 8, 2024) - Kiora Pharmaceuticals, Inc. (NASDAQ: KPRX) ("Kiora" or the "Company") today announced third quarter 2024 financial results and provided an update on its pipeline of therapeutics for the treatment of retinal diseases. Key third quarter and recent corporate highlights include: Investigational new drug application approval to initiate ABACUS-2, the Phase 2 study of... Read More