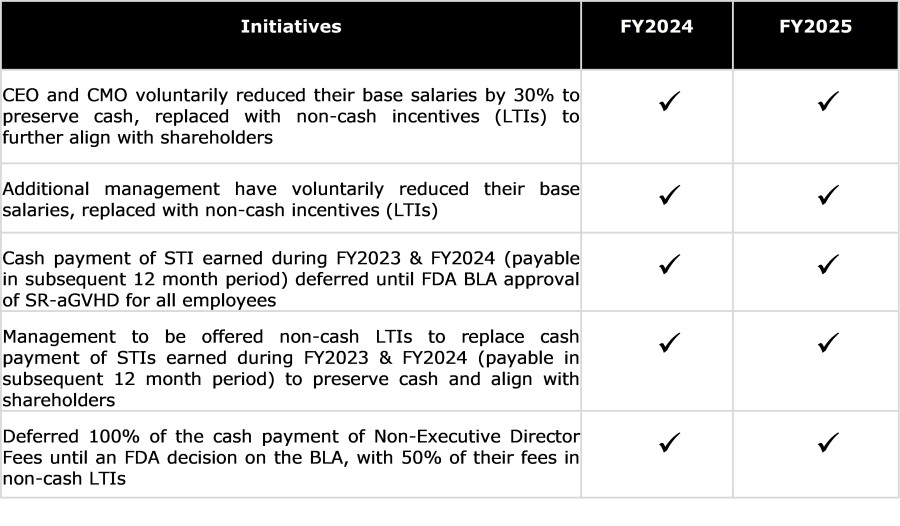
RYONCIL (remestemcel-L) is the first MSC product approved by FDA for any indication. RYONCIL is the first FDA-approved therapy for children aged 2 months and older, including adolescents and teenagers, with steroid-refractory acute graft versus host disease (SR-aGvHD), a life-threatening condition with high mortality rates. In a single-arm, multi-center, Phase 3 trial of children with SR-aGvHD, 89% of whom had high severity... Read More