SAN DIEGO, Oct. 03, 2023 (GLOBE NEWSWIRE) -- ARS Pharmaceuticals, Inc. (Nasdaq: SPRY), a biopharmaceutical company dedicated to empowering at-risk patients and caregivers to better protect patients from severe allergic reactions that could lead to anaphylaxis, announced today the publication of its full pharmacokinetic and pharmacodynamic results from one of its four primary registration studies of neffy (epinephrine nasal spray) in the Journal of Allergy and Clinical Immunology (JACI). JACI is the official scientific publication of the American Academy of Allergy, Asthma and Immunology and the most cited journal in the field of allergy and clinical immunology. The clinical study evaluated single and repeat doses of neffy compared to single and repeat doses of approved injection products in healthy subjects.
Response on PD surrogates of efficacy
The study showed that neffy elicited comparable or greater response compared to approved injection products (intramuscular (IM) injection with needle and syringe, EpiPen), as measured by pharmacodynamic (PD) profile, the surrogate measure for assessing clinical effectiveness in anaphylaxis. These responses in PD were observed even at 1 minute after a single dose of neffy, with mean maximum changes in heart rate (Emax) statistically greater than both IM injection and EpiPen and mean maximum changes in blood pressure (Emax) statistically greater than IM injection and numerically greater than EpiPen. For repeat doses of neffy, mean maximum changes in PD responses for both blood pressure and heart rate were statistically greater than repeat doses of EpiPen.
“Systolic blood pressure (SBP) and heart rate increases result from activation of adrenergic receptors that reverse the anaphylaxis symptoms. Such PD effects occur almost immediately with neffy, which is highly reassuring from a clinical perspective,” said Thomas B. Casale, M.D., Professor of Medicine and Pediatrics and Chief of Clinical and Translational Research in Allergy/Immunology at the University of South Florida in Tampa, and lead author of the study. “Repeat dosing of neffy results in even greater and sustained increases in PD response compared to EpiPen, which could be clinically important in severe cases of anaphylaxis that often need a repeat dose of epinephrine, where patients may experience hypotensive crisis.”
Although the mean PD response for neffy was higher than EpiPen, the absolute maximum changes in blood pressure and heart rate for neffy (i.e., outliers) were comparable to EpiPen and within the range observed during normal activities such as exercise, ensuring neffy PD does not exceed the range observed with approved injection products for safety.
PK profile and safety of neffy
The study also showed that neffy delivered consistent epinephrine levels to attain a pharmacokinetic (PK) profile in the range of approved injection products in healthy adult subjects. In addition, the PK profile for a second dose of neffy was approximately dose-proportional, unlike EpiPen, where the second dose delivered less incremental epinephrine exposure than the first dose. There was also no difference in PK profile between twice dosing of neffy in the same versus different nostrils.
Response on pharmacodynamic (PD) surrogates for efficacy in anaphylaxis for single and twice dosing neffy in healthy subjects.
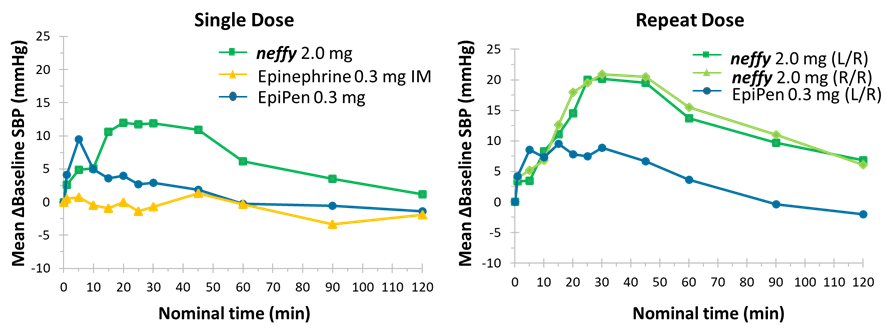 Fig 2 Mean change from baseline in SBP (top), and HR (bottom) vs time. (A) Single dose. (B) Repeat dose.
Fig 2 Mean change from baseline in SBP (top), and HR (bottom) vs time. (A) Single dose. (B) Repeat dose.
neffy was well-tolerated, and all adverse events were mild. As reported previously, the most common adverse events in the neffy 2 mg registration program are mild nasal discomfort (9.7%) and mild headache (6.0%). There is no meaningful pain or irritation as assessed by formal scales with mean VAS scores of 5 to 8 on a scale of 100.
“The single and repeat dose neffy study is part of a rigorous registration program involving more than 600 subjects,” said Dr. Sarina Tanimoto, M.D., Ph.D., Chief Medical Officer and Co-Founder of ARS Pharma. “We’re pleased and grateful to share this positive data, and its recognition by the preeminent allergy journal as JACI’s first epinephrine clinical trial publication in decades. We believe these results support neffy’s clinical profile with robust PK/PD responses that are even greater than injection upon repeat dosing.”
The article was also highlighted in the “Latest Research” section of the American Academy of Allergy, Asthma and Immunology (AAAAI) website, which is the organization that develops treatment guidelines in the United States for severe allergies.
U.S. regulatory status of neffy
In May 2023, an FDA Advisory Committee (the Pulmonary-Allergy Drugs Advisory Committee (PADAC)) concluded a favorable benefit-risk profile of neffy for adults and children (≥30 kg) for the treatment of patients with allergic reactions (Type I), including anaphylaxis. Although no member of the Committee requested a repeat dose study with neffy during allergen-induced allergic rhinitis, and ARS Pharma aligned with FDA in August 2023 that such a study could be completed as a post-marketing requirement, FDA is now requiring ARS Pharma to complete this study prior to approval as outlined in the Complete Response Letter for neffy sent on September 19, 2023.
FDA stated in its PADAC briefing document that rates of any nasal mucosal symptoms, including rhinitis, range from 2% to 11% in anaphylaxis patients, with an average frequency reported in the literature of 5%. After a single dose with neffy during allergen-induced allergic rhinitis, the FDA stated that 73% of patients with moderate to severe symptoms had complete resolution of nasal symptoms by 30 minutes after dosing, due to epinephrine’s well-known decongestant effects (Cleveland Clinic 2023, Epinephrine Nasal Spray).
ARS Pharma plans to complete a repeat dose study with neffy in allergen-induced allergic rhinitis and file its NDA resubmission to the FDA in the first half of 2024, with an anticipated launch of neffy, if approved, in the second half of 2024.
About Type I Allergic Reactions, including Anaphylaxis
Type I severe allergic reactions are serious and potentially life-threatening events that can occur within minutes of exposure to an allergen and require immediate treatment with epinephrine, the only FDA-approved medication for these reactions. While epinephrine autoinjectors have been shown to be highly effective, there are well-published limitations that result in many patients and caregivers delaying or not administering treatment in an emergency situation. These limitations include fear of the needle, lack of portability, needle-related safety concerns, lack of reliability, and complexity of the devices. There are approximately 40 million people in the United States who experience Type I severe allergic reactions due to food, venom, or insect stings. Of those, only 3.3 million currently have an active epinephrine autoinjector prescription, and of those, only half consistently carry their prescribed autoinjector. Even if patients or caregivers carry an autoinjector, more than half either delay or do not administer the device when needed in an emergency.
About ARS Pharmaceuticals, Inc.
ARS Pharma is a biopharmaceutical company dedicated to empowering at-risk patients and caregivers to better protect patients from severe allergic reactions that could lead to anaphylaxis. The Company is developing neffy® (also referred to as ARS-1), an intranasal epinephrine product in clinical development for patients and their caregivers with Type I allergic reactions, including food, medications, and insect bites that could lead to life-threatening anaphylaxis. For more information, visit www.ars-pharma.com.
Forward-Looking Statements
Statements in this press release that are not purely historical in nature are “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995. These statements include, but are not limited to ARS Pharma’s ability to complete the newly required trial and provide the additional information requested by the FDA in the CRL on the timing anticipated, or at all; the potential approval of neffy; the expected timing for the Committee for Medicinal Products for Human Use opinion with respect to the marketing authorization application for neffy; the expected submissions of neffy to other regulatory authorities in additional countries and the timing thereof; ARS Pharma’s cash, cash equivalents and short-term investments on hand upon any future approval of neffy; and other statements that are not historical fact. Because such statements are subject to risks and uncertainties, actual results may differ materially from those expressed or implied by such forward-looking statements. Words such as “anticipate,” “plans,” “expects,” “will,” “potential” and similar expressions are intended to identify forward-looking statements. These forward-looking statements are based upon ARS Pharma’s current expectations and involve assumptions that may never materialize or may prove to be incorrect. Actual results and the timing of events could differ materially from those anticipated in such forward-looking statements as a result of various risks and uncertainties, which include, without limitation, the ability to obtain and maintain regulatory approval for neffy; the ability to successful complete the newly requested trial on the timeframe anticipated at all, as a result of challenges inherent to enrolling, conducting and completing clinical trials; the results of the new clinical trial may not support the approval of neffy; results from clinical trials may not be indicative of results that may be observed in the future; potential safety and other complications from neffy; the labelling for neffy, if approved; the scope, progress and expansion of developing and commercializing neffy; the size and growth of the market therefor and the rate and degree of market acceptance thereof vis-à-vis intramuscular injectable products; ARS Pharma’s ability to protect its intellectual property position; and the impact of government laws and regulations. Additional risks and uncertainties that could cause actual outcomes and results to differ materially from those contemplated by the forward-looking statements are included under the caption “Risk Factors” in ARS Pharma’s Quarterly Report on Form 10-Q for the quarter ended June 30, 2023, filed with the Securities and Exchange Commission on August 10, 2023. This document can also be accessed on ARS Pharma’s web page at ir.ars-pharma.com by clicking on the link “Financials & Filings.”
The forward-looking statements included in this press release are made only as of the date hereof. ARS Pharma assumes no obligation and does not intend to update these forward-looking statements, except as required by law.
ARS Pharma Investor Contact:
Justin Chakma
ARS Pharmaceuticals
This email address is being protected from spambots. You need JavaScript enabled to view it.
ARS Pharma Media Contact:
Laura O'Neill
FINN Partners
This email address is being protected from spambots. You need JavaScript enabled to view it.

| Last Trade: | US$14.01 |
| Daily Change: | -0.10 -0.71 |
| Daily Volume: | 1,516,879 |
| Market Cap: | US$1.360B |
November 13, 2024 October 24, 2024 September 09, 2024 | |

Recursion Pharmaceuticals is a clinical stage TechBio company leading the space by decoding biology to industrialize drug discovery. Enabling its mission is the Recursion OS, a platform built across diverse technologies that continuously expands one of the world’s largest....
CLICK TO LEARN MORE
Chimerix is on a mission to develop medicines that meaningfully improve and extend the lives of patients facing deadly diseases. The company is devoted to filling gaps in the treatment paradigm. Chimerix’s most advanced clinical-stage program is in development for H3 K27M-mutant glioma....
CLICK TO LEARN MOREEnd of content
No more pages to load
COPYRIGHT ©2023 HEALTH STOCKS HUB