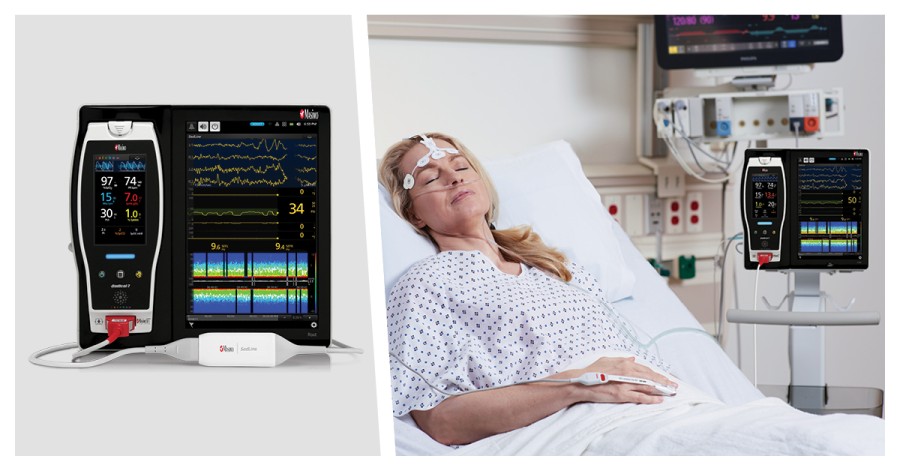
IRVINE, Calif. / Aug 07, 2023 / Business Wire / Masimo (NASDAQ: MASI) today announced the findings of a study published in Frontiers in Neuroscience in which Dr. Lichy Han and colleagues at Stanford University evaluated the ability of Masimo SedLine® brain function monitoring to assess the sedation levels of patients undergoing drug-induced sleep endoscopy (DISE) with dexmedetomidine – a sedative with numerous advantages. The researchers found that Masimo Patient State Index (PSi), a SedLine parameter derived from electroencephalographic (EEG) data, was statistically significantly associated with dexmedetomidine dosage, and, along with another EEG metric, “better captured changes in brain state from dexmedetomidine and ha[s] potential to improve the monitoring of dexmedetomidine sedation.”1
The researchers noted that while anesthesia induced with dexmedetomidine may more closely resemble natural sleep compared to other sedatives, which makes it particularly appropriate in procedures like DISE, its effects on the brain are inconsistent, which has limited its usage. Better sedation assessment – an improved understanding of its impact and corresponding EEG changes – could help support more widespread adoption of the drug. The researchers accordingly sought to compare and evaluate several methods of assessing sedation in such a scenario. Regarding the choice of EEG markers, they noted that “Previous EEG markers, such as the bispectral index, have been unable to determine differences elicited with dexmedetomidine (Kaskinoro et al., 2011), thus motivating the discovery of novel EEG markers associated with changes in brain state seen with dexmedetomidine sedation.”
They enrolled 51 patients undergoing DISE, for the diagnosis and possible treatment of obstructive sleep apnea, who were sedated with dexmedetomidine and continuously monitored using Masimo SedLine. The concentration of the drug in their bloodstream was determined using a pharmacokinetic model. The concentration levels over time were recorded alongside the values derived from four methods of assessment, two metrics and two analyses: 1) the Richmond Agitation-Sedation Scale (RASS), 2) the SedLine Patient State Index (PSi), 3) spectral edge frequency (SEF95), the frequency below which 95% of total EEG power was contained, and 4) the correlation dimension (CD), a type of fractal dimension used to assess the complexity of a system. To evaluate the efficacy of these methods, the researchers compared them both against dexmedetomidine concentration and against each other.
Comparing the assessment methods to dexmedetomidine concentration, the researchers observed that RASS scores decreased with increasing dexmedetomidine concentration but noted that, “Overall, the RASS score was least able to capture the brain state changes seen with dexmedetomidine, suggesting other metrics for titrating dexmedetomidine sedation may be of greater value.” They found that PSi, SEF95, and CD all decreased statistically significantly with increasing dexmedetomidine concentration (p < 0.001, p = 0.006, and p < 0.001, respectively), “a clear dose-dependent decrease with an inflection point.”
Comparing the methods to each other, they found that PSi, SEF95, and CD all decreased statistically significantly with decreasing RASS score (p < 0.001, p < 0.001, and p = 0.02, respectively), most notably at deeper levels of sedation, suggesting that this is when they may be most beneficial. CD and SEF95 were statistically significantly correlated (p < 0.001, r = 0.515), but there was a subset of points that exhibited high SEF95 but low CD. PSi was most strongly correlated with SEF95 (p < 0.001, r = 0.631).
The researchers concluded, “Complex EEG metrics such as PSi and CD, as compared to RASS score and SEF95, better captured changes in brain state from dexmedetomidine and have potential to improve the monitoring of dexmedetomidine sedation.” In their discussion, they noted that, “CD excelled at reflecting changes seen with rising dexmedetomidine concentration but appeared to exhibit a ceiling effect, whereas PSi had the most significant relationship with the RASS score. Both metrics were able to capture brain state changes corresponding to increased dexmedetomidine dose, showed decreased sensitivity to observer variability and artifact, and corresponded to currently used infusion dosages in pharmacokinetic simulations.”
Study lead authors Lichy Han, MD, PhD and David Drover, MD, Perioperative and Pain Medicine at Stanford University, commented, “This study provides one more step in the journey to understanding SedLine processed EEG parameters that can guide sedation levels. In this case, dexmedetomidine, a widely used sedative, which has not been fully characterized by processed EEG, was titrated using standard indices PSi, SEF95, as well as fractal dimension (CD) commonly applied to time series data like EEG. In contrast to BIS, SedLine parameters (particularly PSi) were able to characterize processed EEG changes that correlated with dexmedetomidine sedation level and concentration, and these parameters correlated better than the subjective measure RASS (the most widely used clinical measure of sedation depth). This work helps clinicians use SedLine as an objective measure reflecting how the brain is responding to this important sedative, dexmedetomidine.”
@Masimo | #Masimo
About Masimo
Masimo (NASDAQ: MASI) is a global medical technology company that develops and produces a wide array of industry-leading monitoring technologies, including innovative measurements, sensors, patient monitors, and automation and connectivity solutions. In addition, Masimo Consumer Audio is home to eight legendary audio brands, including Bowers & Wilkins, Denon, Marantz, and Polk Audio. Our mission is to improve life, improve patient outcomes, and reduce the cost of care. Masimo SET® Measure-through Motion and Low Perfusion™ pulse oximetry, introduced in 1995, has been shown in over 100 independent and objective studies to outperform other pulse oximetry technologies.2 Masimo SET® has also been shown to help clinicians reduce severe retinopathy of prematurity in neonates,3 improve CCHD screening in newborns4 and, when used for continuous monitoring with Masimo Patient SafetyNet™ in post-surgical wards, reduce rapid response team activations, ICU transfers, and costs.5-8 Masimo SET® is estimated to be used on more than 200 million patients in leading hospitals and other healthcare settings around the world,9 and is the primary pulse oximetry at 9 of the top 10 hospitals as ranked in the 2022-23 U.S. News and World Report Best Hospitals Honor Roll.10 In 2005, Masimo introduced rainbow® Pulse CO-Oximetry technology, allowing noninvasive and continuous monitoring of blood constituents that previously could only be measured invasively, including total hemoglobin (SpHb®), oxygen content (SpOC™), carboxyhemoglobin (SpCO®), methemoglobin (SpMet®), Pleth Variability Index (PVi®), RPVi™ (rainbow® PVi), and Oxygen Reserve Index (ORi™). In 2013, Masimo introduced the Root® Patient Monitoring and Connectivity Platform, built from the ground up to be as flexible and expandable as possible to facilitate the addition of other Masimo and third-party monitoring technologies; key Masimo additions include Next Generation SedLine® Brain Function Monitoring, O3® Regional Oximetry, and ISA™ Capnography with NomoLine® sampling lines. Masimo’s family of continuous and spot-check monitoring Pulse CO-Oximeters® includes devices designed for use in a variety of clinical and non-clinical scenarios, including tetherless, wearable technology, such as Radius-7®, Radius PPG®, and Radius VSM™, portable devices like Rad-67®, fingertip pulse oximeters like MightySat® Rx, and devices available for use both in the hospital and at home, such as Rad-97®. Masimo hospital and home automation and connectivity solutions are centered around the Masimo Hospital Automation™ platform, and include Iris® Gateway, iSirona™, Patient SafetyNet, Replica®, Halo ION®, UniView®, UniView :60™, and Masimo SafetyNet®. Its growing portfolio of health and wellness solutions includes Radius Tº®, Masimo W1™, and Masimo Stork™. Additional information about Masimo and its products may be found at www.masimo.com. Published clinical studies on Masimo products can be found at www.masimo.com/evidence/featured-studies/feature/.
ORi and RPVi have not received FDA 510(k) clearance and are not available for sale in the United States. The use of the trademark Patient SafetyNet is under license from University HealthSystem Consortium.
References
Forward-Looking Statements
This press release includes forward-looking statements as defined in Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934, in connection with the Private Securities Litigation Reform Act of 1995. These forward-looking statements include, among others, statements regarding the potential effectiveness of Masimo SedLine® and PSi. These forward-looking statements are based on current expectations about future events affecting us and are subject to risks and uncertainties, all of which are difficult to predict and many of which are beyond our control and could cause our actual results to differ materially and adversely from those expressed in our forward-looking statements as a result of various risk factors, including, but not limited to: risks related to our assumptions regarding the repeatability of clinical results; risks related to our belief that Masimo's unique noninvasive measurement technologies, including Masimo SedLine and PSi, contribute to positive clinical outcomes and patient safety; risks that the researchers’ conclusions and findings may be inaccurate; risks related to our belief that Masimo noninvasive medical breakthroughs provide cost-effective solutions and unique advantages; risks related to COVID-19; as well as other factors discussed in the "Risk Factors" section of our most recent reports filed with the Securities and Exchange Commission ("SEC"), which may be obtained for free at the SEC's website at www.sec.gov. Although we believe that the expectations reflected in our forward-looking statements are reasonable, we do not know whether our expectations will prove correct. All forward-looking statements included in this press release are expressly qualified in their entirety by the foregoing cautionary statements. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of today's date. We do not undertake any obligation to update, amend or clarify these statements or the "Risk Factors" contained in our most recent reports filed with the SEC, whether as a result of new information, future events or otherwise, except as may be required under the applicable securities laws.
| Last Trade: | US$169.53 |
| Daily Change: | -1.44 -0.84 |
| Daily Volume: | 120,717 |
| Market Cap: | US$9.080B |
November 26, 2024 November 05, 2024 October 29, 2024 September 25, 2024 | |

Chimerix is on a mission to develop medicines that meaningfully improve and extend the lives of patients facing deadly diseases. The company is devoted to filling gaps in the treatment paradigm. Chimerix’s most advanced clinical-stage program is in development for H3 K27M-mutant glioma....
CLICK TO LEARN MORE
C4 Therapeutics is pioneering a new class of small-molecule drugs that selectively destroy disease-causing proteins via degradation using the innate machinery of the cell. This targeted protein degradation approach offers advantages over traditional drugs, including the potential to treat a wider range of diseases...
CLICK TO LEARN MOREEnd of content
No more pages to load
COPYRIGHT ©2023 HEALTH STOCKS HUB