NEUCHATEL, Switzerland / Aug 21, 2023 / Business Wire / Masimo (NASDAQ: MASI) today announced the findings of a prospective, randomized study published in Perioperative Medicine in which Dr. Yu Wang and colleagues at institutions in Shenzhen, Shaoguan, and Guangzhou, China evaluated the use of noninvasive, continuous Masimo PVi®, as part of goal-directed fluid therapy (GDFT), to guide intraoperative fluid administration during gastrointestinal (GI) surgery on elderly patients by comparing it to conventional fluid therapy. The researchers found that patients in the PVi group had a significantly lower rate of cardiopulmonary complications (8.4% vs 19.2%) and had a significantly lower total volume of fluid administered (median 2075 ml vs. 2500 ml).1
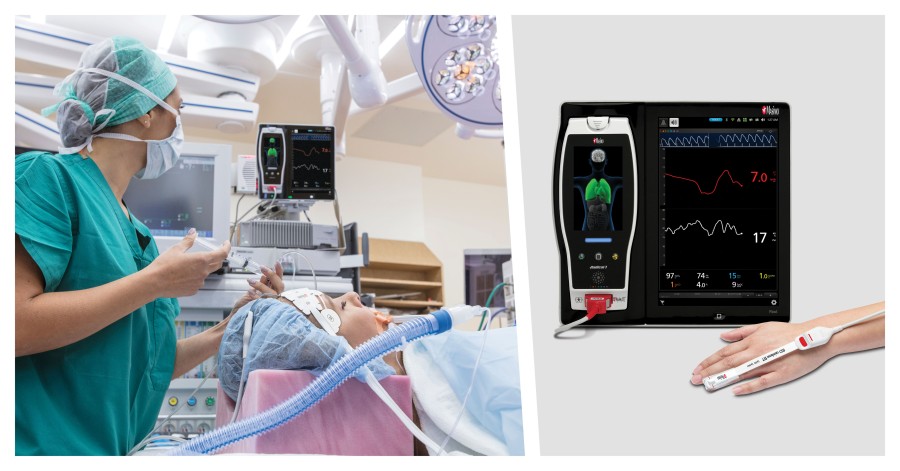
Noting the particular value of optimizing fluid management during GI procedures, especially in elderly patients, because of the high rate of postoperative complications and frequent fluid deficits (because of preoperative fasting, bowel preparation, and intraoperative fluid loss), the researchers sought to determine whether GDFT using Masimo PVi might improve outcomes in this challenging scenario. PVi, or pleth variability index, is a measure of the variations in perfusion index over the respiratory cycle, and has the advantage, compared to arterial line-based methods of gauging fluid responsiveness (e.g. stroke volume variation [SVV] and pulse pressure variation [PPV]), of being obtained via noninvasive Masimo rainbow SET® pulse oximetry and Pulse CO-Oximetry. PVi is indicated as a noninvasive, dynamic indicator of fluid responsiveness in select populations of mechanically ventilated adult patients. As the researchers note, PVi has been shown to perform similarly to invasive methods of fluid assessment, such as PPV and SVV, in a variety of surgeries.2
The researchers enrolled patients aged ≥ 65 years scheduled for elective GI surgery at two university hospitals between November 2017 and December 2020. Patients were randomly assigned to the GDFT group (n = 107) or the conventional fluid therapy (CFT) group (n = 104). In the GDFT group, fluid therapy was guided by PVi obtained from the photoplethysmographic waveform measured by fingertip rainbow® Pulse CO-Oximetry sensors. The outcomes evaluated and compared between the two groups were: composite complications at 30 days after surgery; cardiopulmonary complications at 30 days (pneumonia, atelectatis, pulmonary edema, arrhythmia, and acute myocardial infarction); time to first flatus; postoperative nausea and vomiting; infections including anastomotic leak rates; and postoperative length of hospital stay.
The researchers found that the rate of cardiopulmonary complications, as well as the total volume of fluid administered intraoperatively, were statistically significantly lower in the GDFT (PVi) group, as highlighted in the table below. Although there was a trend toward shorter length of stay and lower anastomotic leakage rates in the PVi group, these and other outcomes were not statistically significantly different.
Outcome | GDFT (PVi) group | CFT group | p-Value* | |
Median volume of. fluids administered (ml) | 2075 (interquartile range: 1900, 2600) | 2500 (2000, 3100) | 0.008 | |
Number (and percentage) of patients with one or more complications | 46 (43%) | 43 (41.3) | 0.089 | |
…with cardiopulmonary complications | 9 (8.4%) | 20 (19.2) | 0.022 | |
…with postop. nausea/vomiting | 42 (39.3%) | 35 (33.7) | 0.398 | |
Median time to first flatus (hours) | 60 (interquartile range: 30, 93) | 52 (34, 81) | 0.475 | |
Number (and percentage) of patients complicated by anastomotic leakage | 1 (0.9%) | 5 (4.8%) | 0.201 | |
Median postop. length of stay (days) | 9 (interquartile range: 8,14) | 10 (8, 12) | 0.614 | |
*A p-value < 0.05 was considered statistically significant. |
|
|
|
The researchers concluded, “Among elderly patients undergoing GI surgery, intraoperative GDFT based on the simple and noninvasive PVi did not reduce the occurrence of composite postoperative complications but was associated with a lower cardiopulmonary complication rate than usual fluid management.”
Joe Kiani, Founder and CEO of Masimo, said, “We introduced PVi in 2007. It was the first and is still the only way of measuring fluid responsiveness noninvasively via pulse oximetry, with our pulse oximeters, at a fraction of the cost of invasive methods and without risk of invasive procedures to the patient. Since then, PVi has made a tremendous contribution to patient care, and its utility as a fluid responsiveness indicator has been shown in more than 100 independent, published studies.2 This latest study adds to the outcomes evidence that PVi can be used to help clinicians manage the fluid levels of their patients without invasive catheters.”
In the U.S., PVi is FDA 510(k) cleared as a noninvasive dynamic indicator of fluid responsiveness in select populations of mechanically ventilated adult patients. Accuracy of PVi in predicting fluid responsiveness is variable and influenced by numerous patient, procedure and device related factors. PVi measures the variation in the plethysmography amplitude but does not provide measurements of stroke volume or cardiac output. Fluid management decisions should be based on a complete assessment of the patient’s condition and should not be based solely on PVi.
@Masimo | #Masimo
About Masimo
Masimo (NASDAQ: MASI) is a global medical technology company that develops and produces a wide array of industry-leading monitoring technologies, including innovative measurements, sensors, patient monitors, and automation and connectivity solutions. In addition, Masimo Consumer Audio is home to eight legendary audio brands, including Bowers & Wilkins, Denon, Marantz, and Polk Audio. Our mission is to improve life, improve patient outcomes, and reduce the cost of care. Masimo SET® Measure-through Motion and Low Perfusion™ pulse oximetry, introduced in 1995, has been shown in over 100 independent and objective studies to outperform other pulse oximetry technologies.3 Masimo SET® has also been shown to help clinicians reduce severe retinopathy of prematurity in neonates,4 improve CCHD screening in newborns5 and, when used for continuous monitoring with Masimo Patient SafetyNet™ in post-surgical wards, reduce rapid response team activations, ICU transfers, and costs.6-9 Masimo SET® is estimated to be used on more than 200 million patients in leading hospitals and other healthcare settings around the world,10 and is the primary pulse oximetry at 9 of the top 10 hospitals as ranked in the 2022-23 U.S. News and World Report Best Hospitals Honor Roll.11 In 2005, Masimo introduced rainbow® Pulse CO-Oximetry technology, allowing noninvasive and continuous monitoring of blood constituents that previously could only be measured invasively, including total hemoglobin (SpHb®), oxygen content (SpOC™), carboxyhemoglobin (SpCO®), methemoglobin (SpMet®), Pleth Variability Index (PVi®), RPVi™ (rainbow® PVi), and Oxygen Reserve Index (ORi™). In 2013, Masimo introduced the Root® Patient Monitoring and Connectivity Platform, built from the ground up to be as flexible and expandable as possible to facilitate the addition of other Masimo and third-party monitoring technologies; key Masimo additions include Next Generation SedLine® Brain Function Monitoring, O3® Regional Oximetry, and ISA™ Capnography with NomoLine® sampling lines. Masimo’s family of continuous and spot-check monitoring Pulse CO-Oximeters® includes devices designed for use in a variety of clinical and non-clinical scenarios, including tetherless, wearable technology, such as Radius-7®, Radius PPG®, and Radius VSM™, portable devices like Rad-67®, fingertip pulse oximeters like MightySat® Rx, and devices available for use both in the hospital and at home, such as Rad-97®. Masimo hospital and home automation and connectivity solutions are centered around the Masimo Hospital Automation™ platform, and include Iris® Gateway, iSirona™, Patient SafetyNet, Replica®, Halo ION®, UniView®, UniView :60™, and Masimo SafetyNet®. Its growing portfolio of health and wellness solutions includes Radius Tº®, Masimo W1™, and Masimo Stork™. Additional information about Masimo and its products may be found at www.masimo.com. Published clinical studies on Masimo products can be found at www.masimo.com/evidence/featured-studies/feature/.
ORi and RPVi have not received FDA 510(k) clearance and are not available for sale in the United States. The use of the trademark Patient SafetyNet is under license from University HealthSystem Consortium.
References
Forward-Looking Statements
This press release includes forward-looking statements as defined in Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934, in connection with the Private Securities Litigation Reform Act of 1995. These forward-looking statements include, among others, statements regarding the potential effectiveness of Masimo PVi®. These forward-looking statements are based on current expectations about future events affecting us and are subject to risks and uncertainties, all of which are difficult to predict and many of which are beyond our control and could cause our actual results to differ materially and adversely from those expressed in our forward-looking statements as a result of various risk factors, including, but not limited to: risks related to our assumptions regarding the repeatability of clinical results; risks related to our belief that Masimo's unique noninvasive measurement technologies, including Masimo PVi, contribute to positive clinical outcomes and patient safety; risks that the researchers’ conclusions and findings may be inaccurate; risks related to our belief that Masimo noninvasive medical breakthroughs provide cost-effective solutions and unique advantages; risks related to COVID-19; as well as other factors discussed in the "Risk Factors" section of our most recent reports filed with the Securities and Exchange Commission ("SEC"), which may be obtained for free at the SEC's website at www.sec.gov. Although we believe that the expectations reflected in our forward-looking statements are reasonable, we do not know whether our expectations will prove correct. All forward-looking statements included in this press release are expressly qualified in their entirety by the foregoing cautionary statements. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of today's date. We do not undertake any obligation to update, amend or clarify these statements or the "Risk Factors" contained in our most recent reports filed with the SEC, whether as a result of new information, future events or otherwise, except as may be required under the applicable securities laws.
| Last Trade: | US$169.52 |
| Daily Change: | -0.48 -0.28 |
| Daily Volume: | 796,263 |
| Market Cap: | US$9.080B |
November 26, 2024 November 05, 2024 October 29, 2024 September 25, 2024 | |

C4 Therapeutics is pioneering a new class of small-molecule drugs that selectively destroy disease-causing proteins via degradation using the innate machinery of the cell. This targeted protein degradation approach offers advantages over traditional drugs, including the potential to treat a wider range of diseases...
CLICK TO LEARN MORE
Cue Biopharma is developing the first-ever class of therapeutics for the treatment of cancer that mimic the natural signals, or “Cues”, of the immune system. This novel class of injectable biologics selectively engages and modulates tumor-specific T cells directly within the patient’s body to transform...
CLICK TO LEARN MOREEnd of content
No more pages to load
COPYRIGHT ©2023 HEALTH STOCKS HUB