SAN FRANCISCO / Oct 13, 2023 / Business Wire / Masimo (NASDAQ: MASI) today announced that ORi™, a noninvasive, continuous parameter designed to provide additional insight into a patient’s oxygen status in the moderate hyperoxic range under supplemental oxygen, has been granted a De Novo by the FDA. Enabled by the multi-wavelength Masimo rainbow® Pulse CO-Oximetry platform, ORi is designed for use in conjunction with oxygen saturation (SpO2) to provide increased resolution of changes in oxygenation under supplemental oxygen. With the De Novo, ORi becomes the first-of-its-kind parameter cleared by the FDA to help clinicians manage oxygen of adults undergoing surgery in perioperative hospital environments.
Without ORi, there is no noninvasive way to monitor oxygenation under supplemental oxygen to manage hyperoxia, or higher than normal oxygenation of arterial blood. There is growing evidence that hyperoxia is harmful and can lead to oxygen toxicity, causing oxygen poisoning or pulmonary tissue damage.1 Currently, clinicians take blood draws that are analyzed to determine PaO2 levels, the partial pressure of oxygen measured by arterial blood gas devices. However, arterial blood analyses are both intermittent and delayed – leaving clinicians blind to the changes in oxygenation occurring between blood draw results.
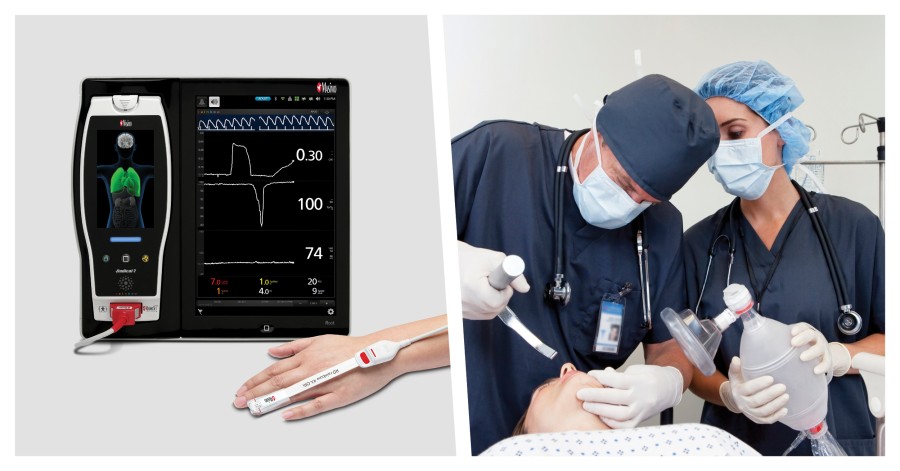
Masimo ORi addresses these shortcomings by providing continuous insight into the oxygenation of hemoglobin in the moderate hyperoxic range (PaO2 > 100 and ≤ 250 mmHg). ORi is trended continuously with SpO2 as a unit-less index between 0.00 and 1.00 to extend the visibility of the oxygenation of patients beyond SpO2 under supplemental oxygen. By convention, SpO2 is limited to an upper limit of 100%, but oxygenation can rise into hyperoxia when supplemental oxygen is administered. ORi provides clinicians with additional visibility, as a complement to Masimo SET® pulse oximetry, into when oxygenation is increased into, or decreased out of, moderate hyperoxia, in real time.
Numerous studies have demonstrated ORi’s utility. For example, in a study published in Anesthesia & Analgesia of 106 adult patients undergoing scheduled surgery, researchers found decreases in ORi “may provide advance indication of falling PaO2 when SpO2 is still > 98%.”2 In another study published in Intensive Care Medicine, researchers found that the use of ORi monitoring to titrate oxygen rates “allowed an important reduction of the time spent with hyperoxia compared with the use of SpO2 alone,” in a group of 150 mechanically ventilated adult patients randomized to an ORi or a control group.3
Joe Kiani, Founder and CEO of Masimo, said, “Since ORi’s availability and success outside the U.S., perioperative clinicians in the U.S. have been waiting for a way to noninvasively monitor patients under supplemental oxygen beyond the limits of SpO2. We are thrilled that U.S. clinicians can now integrate ORi monitoring – available now on our rainbow SET® platform – into their oxygenation monitoring practices, alongside Masimo SET® Measure through Motion and Low Perfusion™ pulse oximetry, and experience their combined benefits.”
With the De Novo, Masimo is introducing a new sensor line to the market, RD rainbow™ 4λ sensors, expanding the RD family of pulse CO-oximetry sensors, which are now available in four levels of capabilities:
Jesse M. Ehrenfeld, MD, President of the American Medical Association and advisor to Masimo, commented, “I can envision a number of scenarios I encounter in my daily clinical practice as an anesthesiologist where ORi would be invaluable. During patient pre-oxygenation, I often find myself unsure of the adequacy of pre-oxygenation, especially in a patient with a significant cardiopulmonary comorbidity or a patient with diminished oxygen reserve capacity. ORi would solve this problem by giving an easy-to-understand parameter that provides visibility to how oxygenation is changing during the pre-oxygenation process. Additionally, I often find that during the management of a difficult airway, it is never quite clear when to stop, re-establish mask ventilation, and allow the patient to recover. Again, ORi would be very helpful in addressing this issue as it can track the trajectory of the patient’s oxygenation status. I am delighted to see ORi receive FDA clearance. It is not often that new parameters are developed which can actually make a real impact in clinical practice.”
Richard L. Applegate II, MD, Chair of Anesthesiology at Loma Linda University Health, California, stated, “Our studies show that ORi provides advanced detection of low SpO2 events. This additional time may allow modification of airway management, earlier calls for help, or assistance from other providers. Advanced detection of worsening oxygenation is valuable in operative and critical care settings and ORi use has the potential to provide continuous monitoring to detect changes in pulmonary function.”
Ken B. Johnson, MD, Professor of Anesthesiology, University of Utah, commented, “I have been eager for the ORi parameter to be available in the U.S. for a long time as it is a unique innovation with the potential to significantly improve patient care. Observing the ORi trend may help clinicians anticipate hemoglobin oxygen desaturations before they occur. Clinical scenarios where this technology may be useful include airway management that takes longer than expected, prompting rescue breaths before desaturation occurs, and procedural sedation with unanticipated prolonged periods of apnea that can trigger rescue maneuvers before the onset of unwanted desaturation. This index in combination with pulse oximetry shows promise in better managing adverse events related to poor oxygenation and improved patient outcomes.”
ORi is granted a De Novo by the US FDA to be used in patients undergoing surgery as an adjunct to SpO2 for increased monitoring resolution of elevated hemoglobin oxygen saturation levels (e.g., due to administration of supplemental oxygen).
The ORi feature is indicated for the monitoring of hemoglobin oxygen saturation levels in patients 18 years and older (adults and transitional adolescents) on supplemental oxygen during no-motion conditions perioperatively in hospital environments.
*SpfO2 and RD rainbow 12λ sensors have obtained CE Marking. Not available in the U.S.
@Masimo | #Masimo
About Masimo
Masimo (NASDAQ: MASI) is a global medical technology company that develops and produces a wide array of industry-leading monitoring technologies, including innovative measurements, sensors, patient monitors, and automation and connectivity solutions. In addition, Masimo Consumer Audio is home to eight legendary audio brands, including Bowers & Wilkins, Denon, Marantz, and Polk Audio. Our mission is to improve life, improve patient outcomes, and reduce the cost of care. Masimo SET® Measure-through Motion and Low Perfusion™ pulse oximetry, introduced in 1995, has been shown in over 100 independent and objective studies to outperform other pulse oximetry technologies.4 Masimo SET® has also been shown to help clinicians reduce severe retinopathy of prematurity in neonates,5 improve CCHD screening in newborns6 and, when used for continuous monitoring with Masimo Patient SafetyNet™ in post-surgical wards, reduce rapid response team activations, ICU transfers, and costs.7-10 Masimo SET® is estimated to be used on more than 200 million patients in leading hospitals and other healthcare settings around the world,11 and is the primary pulse oximetry at 9 of the top 10 hospitals as ranked in the 2022-23 U.S. News and World Report Best Hospitals Honor Roll.12 In 2005, Masimo introduced rainbow® Pulse CO-Oximetry technology, allowing noninvasive and continuous monitoring of blood constituents that previously could only be measured invasively, including total hemoglobin (SpHb®), oxygen content (SpOC™), carboxyhemoglobin (SpCO®), methemoglobin (SpMet®), Pleth Variability Index (PVi®), RPVi™ (rainbow® PVi), and Oxygen Reserve Index (ORi™). In 2013, Masimo introduced the Root® Patient Monitoring and Connectivity Platform, built from the ground up to be as flexible and expandable as possible to facilitate the addition of other Masimo and third-party monitoring technologies; key Masimo additions include Next Generation SedLine® Brain Function Monitoring, O3® Regional Oximetry, and ISA™ Capnography with NomoLine® sampling lines. Masimo’s family of continuous and spot-check monitoring Pulse CO-Oximeters® includes devices designed for use in a variety of clinical and non-clinical scenarios, including tetherless, wearable technology, such as Radius-7®, Radius PPG®, and Radius VSM™, portable devices like Rad-67®, fingertip pulse oximeters like MightySat® Rx, and devices available for use both in the hospital and at home, such as Rad-97®. Masimo hospital and home automation and connectivity solutions are centered around the Masimo Hospital Automation™ platform, and include Iris® Gateway, iSirona™, Patient SafetyNet, Replica®, Halo ION®, UniView®, UniView :60™, and Masimo SafetyNet®. Its growing portfolio of health and wellness solutions includes Radius Tº®, Masimo W1™, and Masimo Stork™. Additional information about Masimo and its products may be found at www.masimo.com. Published clinical studies on Masimo products can be found at www.masimo.com/evidence/featured-studies/feature/.
RPVi has not received FDA 510(k) clearance and is not available for sale in the United States. The use of the trademark Patient SafetyNet is under license from University HealthSystem Consortium.
References
Forward-Looking Statements
This press release includes forward-looking statements as defined in Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934, in connection with the Private Securities Litigation Reform Act of 1995. These forward-looking statements include, among others, statements regarding the potential effectiveness of Masimo ORi™ and RD rainbow™ sensors. These forward-looking statements are based on current expectations about future events affecting us and are subject to risks and uncertainties, all of which are difficult to predict and many of which are beyond our control and could cause our actual results to differ materially and adversely from those expressed in our forward-looking statements as a result of various risk factors, including, but not limited to: risks related to our assumptions regarding the repeatability of clinical results; risks related to our belief that Masimo's unique noninvasive measurement technologies, including Masimo ORi and RD rainbow sensors, contribute to positive clinical outcomes and patient safety; risks related to our belief that Masimo noninvasive medical breakthroughs provide cost-effective solutions and unique advantages; risks related to COVID-19; as well as other factors discussed in the "Risk Factors" section of our most recent reports filed with the Securities and Exchange Commission ("SEC"), which may be obtained for free at the SEC's website at www.sec.gov. Although we believe that the expectations reflected in our forward-looking statements are reasonable, we do not know whether our expectations will prove correct. All forward-looking statements included in this press release are expressly qualified in their entirety by the foregoing cautionary statements. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of today's date. We do not undertake any obligation to update, amend or clarify these statements or the "Risk Factors" contained in our most recent reports filed with the SEC, whether as a result of new information, future events or otherwise, except as may be required under the applicable securities laws.
| Last Trade: | US$169.52 |
| Daily Change: | -0.48 -0.28 |
| Daily Volume: | 796,263 |
| Market Cap: | US$9.080B |
November 26, 2024 November 05, 2024 October 29, 2024 September 25, 2024 | |

Recursion Pharmaceuticals is a clinical stage TechBio company leading the space by decoding biology to industrialize drug discovery. Enabling its mission is the Recursion OS, a platform built across diverse technologies that continuously expands one of the world’s largest....
CLICK TO LEARN MORE
Cue Biopharma is developing the first-ever class of therapeutics for the treatment of cancer that mimic the natural signals, or “Cues”, of the immune system. This novel class of injectable biologics selectively engages and modulates tumor-specific T cells directly within the patient’s body to transform...
CLICK TO LEARN MOREEnd of content
No more pages to load
COPYRIGHT ©2023 HEALTH STOCKS HUB