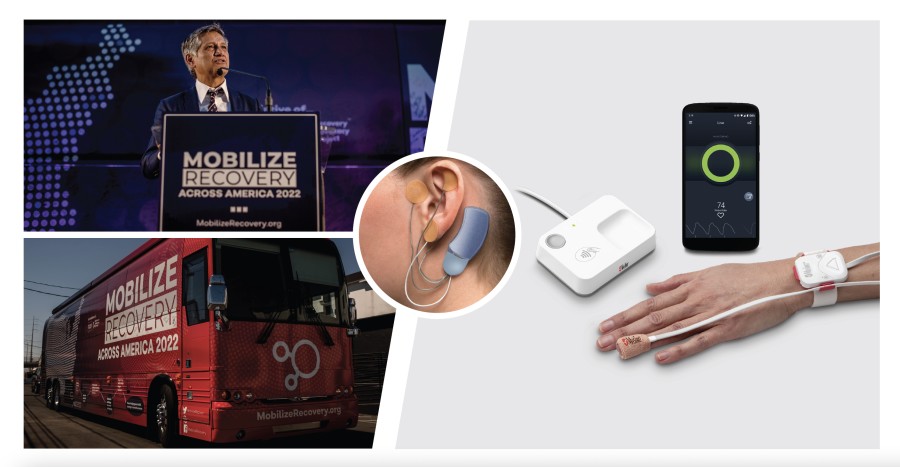
IRVINE, Calif. / Jun 05, 2024 / Business Wire / Masimo (NASDAQ: MASI), a leading global medical technology innovator, today announced that it is partnering with Mobilize Recovery, a nonprofit organization dedicated to ending America’s addiction and overdose crisis, by sponsoring and joining the Mobilize Recovery 2024 bus tour. The tour, which kicks off on September 20th in Los Angeles, with the first stop in Irvine, at the Masimo headquarters, is set to travel to 15 key cities around the U.S. to raise awareness, educate, and support communities around the country as they seek to prevent overdose and make sustainable improvements in addiction recovery outcomes. As part of this sixth annual initiative, Mobilize Recovery and Masimo will be showcasing two key Masimo innovations, Opioid Halo™ and Bridge™, which are designed to help people who are using opioids stay safe and reduce withdrawal symptoms during addiction recovery, respectively.
Founded by Mr. Ryan Hampton, Mobilize Recovery brings together recovery advocates, nonprofit organizations, allies, business leaders and innovators like Masimo, government partners, and like-minded community-based organizations with an interest in creating sustainable change and community solutions, as well as celebrating recovery from substance abuse disorder and mental health challenges. Engaging year-round with mobilizers, Mobilize Recovery provides partnership support and education to uplift and sustain partners’ work; help them develop action-oriented, measurable, scalable goals; raise public awareness; and prevent overdose.
Ryan Hampton, Founder and Executive Director of Mobilize Recovery, said, “Mobilize Recovery champions a grassroots approach, empowering advocates, innovators, businesses, and communities battling the overdose crisis firsthand. We see Masimo as a leading innovation partner in the recovery movement, uniquely positioned to drive technological advancements that make a real impact. We're thrilled to collaborate with a company so committed to leveraging technology for groundbreaking solutions in recovery. Masimo's Opioid Halo and Bridge have shown immense promise in helping opioid users, and we're eager to see their full potential realized. In Masimo’s CEO, Joe Kiani, we've found a passionate voice advocating for overcoming this epidemic, a true thought leader in the recovery field with an unwavering dedication to making a difference.”
Joe Kiani, Founder and CEO of Masimo, added, “We are excited to sponsor Mobilize Recovery, partner with their amazing team, and help kick off the bus tour in September. Masimo is committed to creating evidence-based solutions that fill in existing gaps in patient support networks. We were honored to be chosen by the FDA as a winner of their FDA Opioid Innovation Challenge for our solution to help prevent opioid overdose. With Opioid Halo, we became the first winner to have an authorized device to address the crisis. Bridge, in turn, is the first FDA-cleared medical device to help in the reduction of opioid withdrawal symptoms. Together, we believe these products, and the amazing efforts of Mobilize Recovery and its partners, can make a real difference in fighting the opioid crisis – as well as help eliminate the stigma around addiction.”
Opioid Halo, an opioid overdose prevention and alert system, was granted a De Novo by the FDA in 2023, making it the first and only FDA-cleared monitoring solution for detecting opioid-induced respiratory depression (OIRD). Opioid Halo advances the forefront of continuous monitoring through its unique Opioid Halo engine, an advanced pattern recognition algorithm which helps detect and quantify the risk of severe OIRD. Combined with its innovative distributed architecture, Opioid Halo helps to manage and send escalating alarms to family members, friends, and caregivers, notifying them that help may be needed due to an opioid overdose – including triggering an automatic wellness call, which may lead to EMS being dispatched.
Bridge, a drug-free opioid withdrawal device, uses neuromodulation to aid in the reduction of symptoms associated with opioid withdrawal. Bridge, which has also been granted a De Novo by the FDA, is the first evidence-based, drug-free, non-surgical device of its kind. While medication-assisted treatment can be effective in helping treat opioid withdrawal symptoms, Bridge may help significantly reduce those symptoms, within 30 minutes, helping users make progress in their treatment by bridging the gap on the road to recovery. Bridge fits behind the ear – applied by a healthcare provider in a short, non-surgical procedure – and works by sending electrical impulses to the nerves around the ear, which transmit them to the brain, providing up to five days of continuous relief from withdrawal symptoms.
@Masimo | #Masimo
About Masimo
Masimo (NASDAQ: MASI) is a global medical technology company that develops and produces a wide array of industry-leading monitoring technologies, including innovative measurements, sensors, patient monitors, and automation and connectivity solutions. In addition, Masimo Consumer Audio is home to eight legendary audio brands, including Bowers & Wilkins, Denon, Marantz, and Polk Audio. Our mission is to improve life, improve patient outcomes, and reduce the cost of care. Masimo SET® Measure-through Motion and Low Perfusion™ pulse oximetry, introduced in 1995, has been shown in over 100 independent and objective studies to outperform other pulse oximetry technologies.1 Masimo SET® has also been shown to help clinicians reduce severe retinopathy of prematurity in neonates,2 improve CCHD screening in newborns,3 and, when used for continuous monitoring with Masimo Patient SafetyNet™ in post-surgical wards, reduce rapid response team activations, ICU transfers, and costs.4-7 Masimo SET® is estimated to be used on more than 200 million patients in leading hospitals and other healthcare settings around the world,8 and is the primary pulse oximetry at 9 of the top 10 hospitals as ranked in the 2022-23 U.S. News and World Report Best Hospitals Honor Roll.9 In 2005, Masimo introduced rainbow® Pulse CO-Oximetry technology, allowing noninvasive and continuous monitoring of blood constituents that previously could only be measured invasively, including total hemoglobin (SpHb®), oxygen content (SpOC™), carboxyhemoglobin (SpCO®), methemoglobin (SpMet®), Pleth Variability Index (PVi®), RPVi™ (rainbow® PVi), and Oxygen Reserve Index (ORi™). In 2013, Masimo introduced the Root® Patient Monitoring and Connectivity Platform, built from the ground up to be as flexible and expandable as possible to facilitate the addition of other Masimo and third-party monitoring technologies; key Masimo additions include Next Generation SedLine® Brain Function Monitoring, O3® Regional Oximetry, and ISA™ Capnography with NomoLine® sampling lines. Masimo’s family of continuous and spot-check monitoring Pulse CO-Oximeters® includes devices designed for use in a variety of clinical and non-clinical scenarios, including tetherless, wearable technology, such as Radius-7®, Radius PPG®, and Radius VSM™, portable devices like Rad-67®, fingertip pulse oximeters like MightySat® Rx, and devices available for use both in the hospital and at home, such as Rad-97® and the Masimo W1® Medical Watch. Masimo hospital and home automation and connectivity solutions are centered around the Masimo Hospital Automation™ platform, and include Iris® Gateway, iSirona™, Patient SafetyNet, Replica®, Halo ION®, UniView®, UniView :60™, and Masimo SafetyNet®. Its growing portfolio of health and wellness solutions includes Radius Tº® and Masimo W1 Sport. Additional information about Masimo and its products may be found at www.masimo.com. Published clinical studies on Masimo products can be found at www.masimo.com/evidence/featured-studies/feature/.
RPVi has not received FDA 510(k) clearance and is not available for sale in the United States. The use of the trademark Patient SafetyNet is under license from University HealthSystem Consortium.
References
Forward-Looking Statements
This press release includes forward-looking statements as defined in Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934, in connection with the Private Securities Litigation Reform Act of 1995. These forward-looking statements include, among others, statements regarding the potential effectiveness of Masimo Opioid Halo™ and Bridge™, as well as the potential benefits of Masimo’s partnership with Mobilize Recovery. These forward-looking statements are based on current expectations about future events affecting us and are subject to risks and uncertainties, all of which are difficult to predict and many of which are beyond our control and could cause our actual results to differ materially and adversely from those expressed in our forward-looking statements as a result of various risk factors, including, but not limited to: risks related to our assumptions regarding the repeatability of clinical results; risks related to our belief that Masimo's unique noninvasive measurement technologies, including Opioid Halo and Bridge, contribute to positive clinical outcomes and patient safety; risks related to our belief that Masimo noninvasive medical breakthroughs provide cost-effective solutions and unique advantages; risks related to COVID-19; as well as other factors discussed in the "Risk Factors" section of our most recent reports filed with the Securities and Exchange Commission ("SEC"), which may be obtained for free at the SEC's website at www.sec.gov. Although we believe that the expectations reflected in our forward-looking statements are reasonable, we do not know whether our expectations will prove correct. All forward-looking statements included in this press release are expressly qualified in their entirety by the foregoing cautionary statements. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of today's date. We do not undertake any obligation to update, amend or clarify these statements or the "Risk Factors" contained in our most recent reports filed with the SEC, whether as a result of new information, future events or otherwise, except as may be required under the applicable securities laws.
| Last Trade: | US$169.52 |
| Daily Change: | -0.48 -0.28 |
| Daily Volume: | 796,263 |
| Market Cap: | US$9.080B |
November 26, 2024 November 05, 2024 October 29, 2024 September 25, 2024 | |

Astria Therapeutics is a biopharmaceutical company, and our mission is to bring life-changing therapies to patients and families affected by rare and niche allergic and immunological diseases. Our lead program, STAR-0215, is a monoclonal antibody inhibitor of plasma kallikrein in clinical development...
CLICK TO LEARN MORE
Recursion Pharmaceuticals is a clinical stage TechBio company leading the space by decoding biology to industrialize drug discovery. Enabling its mission is the Recursion OS, a platform built across diverse technologies that continuously expands one of the world’s largest....
CLICK TO LEARN MOREEnd of content
No more pages to load
COPYRIGHT ©2023 HEALTH STOCKS HUB