FLORHAM PARK, N.J., Sept. 10, 2024 (GLOBE NEWSWIRE) -- BeyondSpring Inc. (NASDAQ: BYSI) (“BeyondSpring” or the “Company”), a clinical-stage global biopharmaceutical company focused on developing innovative cancer therapies, today announces Dublin-3 final phase 3 efficacy data of its late clinical-stage agent Plinabulin in combination with docetaxel in second/third line (2L/3L) advanced and metastatic non-small cell lung cancer (NSCLC) with epidermal growth factor receptor (EGFR) wild-type. The lead principal investigator, Dr. Trevor Feinstein from Piedmont Cancer Institute, presented the data in an oral presentation (OA08.04) at ISLAC 2024 World Conference on Lung Cancer on September 9th 2024 in San Diego, CA. Concurrently, Dublin-3 study was published in the Lancet Respiratory Medicine (https://doi.org/10.1016/S2213-2600(24)00178-4).
Docetaxel remains the standard of care for patients with 2L/3L NSCLC without targetable alterations despite severe neutropenia (>40%) that greatly impair patients’ quality of life. Six recent phase 3 studies in patients with EGFR wild-type NSCLC who were previously treated with immune checkpoint inhibitors failed to show overall survival (OS) benefit compared with docetaxel (SAPPHIRE, LEAP-008, CONTACT01, EVOKE-01, CARMEN-LC03, and CANOPY-2). Two phase 3 studies (LUNAR and TROPION-Lung01) showed positive but mixed outcomes compared with docetaxel.
The DUBLIN-3 study was a multicenter, randomized, single-blind, placebo-controlled trial that enrolled patients from 58 medical centers internationally. For the intention-to-treat (ITT) population, 559 patients received either docetaxel plus plinabulin (n=278 [male 199, female 79]) or docetaxel plus placebo (n=281 [male 207, female 74]).
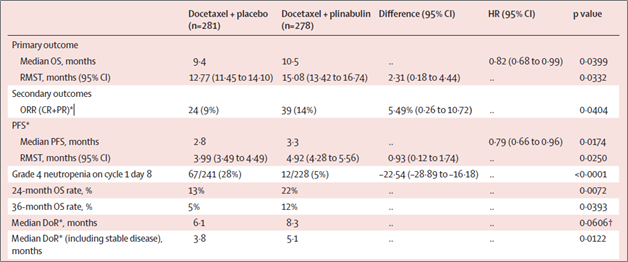
Key findings of Dublin-3 study are summarized below:
“There is a poor prognosis for NSCLC patients without targetable alterations whose disease has progressed on platinum-based therapies. Unfortunately, multiple high-profile phase 3 studies failed to show overall survival benefit in this hard-to-treat population compared to standard of care docetaxel, a drug approved over 20 years ago. The data from DUBLIN-3 study demonstrates that the addition and proper sequencing of plinabulin to docetaxel has a favorable benefit/risk ratio compared with docetaxel alone and may have broad utility. This combination could be considered as a new treatment option for this population with high unmet medical needs,” said Dr. Feinstein, a lead principal investigator of the DUBLIN-3 study at Piedmont Cancer Center, Atlanta.
Citation:
Han B., et al. Plinabulin plus docetaxel versus docetaxel in patients with non-small-cell lung cancer after disease progression on platinum-based regimen (DUBLIN-3): a phase 3, international, multicentre, single-blind, parallel group, randomised controlled trial. Lancet Respir Med. 2024 Sep 09: S2213-2600(24)00178-4.
About Plinabulin
Plinabulin is a novel first-in-class dendritic cell maturation agent with durable anti-cancer benefit observed across multiple clinical studies. As a reversible binder at a distinct tubulin pocket, plinabulin does not change tubulin dynamics or antagonize tubulin stabilizing agents, such as docetaxel, which contributes to its differentiated activity and tolerability compared to other tubulin binders. In addition, plinabulin significantly reduces chemotherapy induced neutropenia and could thereby increase docetaxel tolerability. Over 700 patients have been treated with plinabulin with good tolerability.
About Dublin-3 Study
DUBLIN-3 is a multicenter, single-blinded (patient) and randomized, phase 3 trial in 58 medical centers (US, China, and Australia). Only patients with EGFR wild-type NSCLC who had progressed after first-line platinum-based therapy were enrolled. Patients were randomized (1:1) to receive docetaxel (75 mg/m2) on Day 1 and either plinabulin (30 mg/m2) or placebo on Days 1 and 8 in 21-day cycles until progression, unacceptable toxicity, withdrawal, or death. Treated patients were included in the safety analysis and ITT population in the primary efficacy analyses (NCT02504489). The primary endpoint for the study was OS, and secondary endpoints were PFS, ORR, Duration of Response (DoR), Grade 4 neutropenia and Quality of Life.
BeyondSpring is a global clinical-stage biopharmaceutical company focused on developing innovative therapies to improve clinical outcomes for patients with high unmet medical needs. The Company is advancing its first-in-class lead asset, Plinabulin, a potent inducer of dendritic cell maturation, in late-stage clinical development as a direct anti-cancer agent in NSCLC and a variety of cancer indications. BeyondSpring’s pipeline also includes three preclinical immuno-oncology assets. Additionally, BeyondSpring is an equity owner of SEED Therapeutics, Inc which is a pioneer in Target Protein Degradation technology and its application in innovative drug development. Learn more by visiting https://beyondspringpharma.com.
Investor Contact:
This email address is being protected from spambots. You need JavaScript enabled to view it.
Media Contact:
This email address is being protected from spambots. You need JavaScript enabled to view it.

| Last Trade: | US$2.14 |
| Daily Change: | -0.03 -1.29 |
| Daily Volume: | 19,420 |
| Market Cap: | US$86.320M |
September 16, 2024 | |

Immix Biopharma is a clinical-stage biopharmaceutical company pioneering a novel class of CAR-T cell therapies and Tissue-Specific Therapeutics targeting oncology and immuno-dysregulated diseases with >75 patients treated to-date. Our lead cell therapy asset is NXC-201...
CLICK TO LEARN MORE
Terns Pharmaceuticals is a clinical-stage biopharmaceutical company developing a portfolio of small-molecule product candidates to address serious diseases, including oncology and obesity. Terns’ pipeline contains three clinical stage development programs including GLP-1 receptor...
CLICK TO LEARN MOREEnd of content
No more pages to load
COPYRIGHT ©2023 HEALTH STOCKS HUB