LEXINGTON, Mass. / Apr 25, 2024 / Business Wire / Aldeyra Therapeutics, Inc. (Nasdaq: ALDX) (Aldeyra) will host the Aldeyra 2024 Research & Development Day with investors and financial analysts in New York City to present recent pipeline developments relating to the RASP modulation platform and ADX-2191 for the treatment of retinitis pigmentosa.
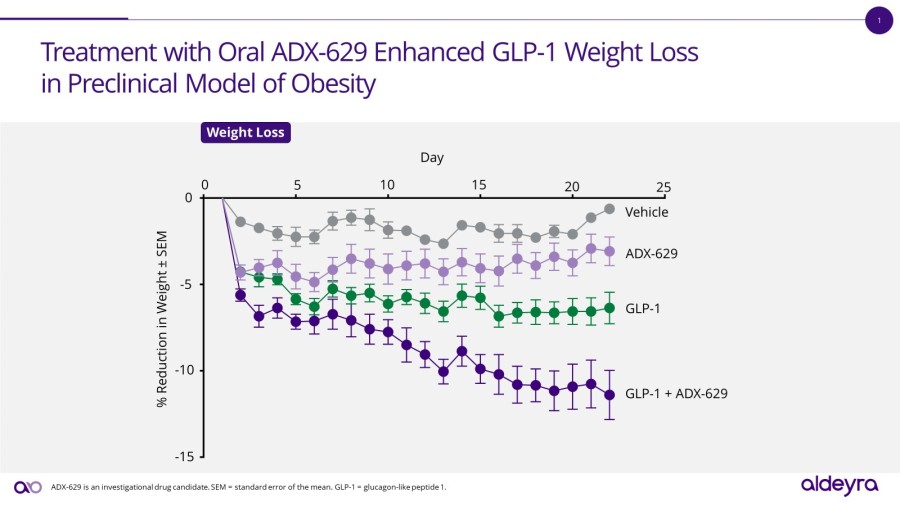
Aldeyra will present new preclinical data from investigational RASP modulators in animal models for obesity, atopic dermatitis, inflammatory pain, and alcoholic hepatitis. In the diet-induced model of obesity, ADX-629 decreased weight and fat mass alone and in combination with a GLP-1 agonist. In the oxazolone model of atopic dermatitis, RASP modulators ADX-629, ADX-246, and ADX-248 demonstrated activity in reducing skin thickness and erosion, and in reducing spleen to body weight ratio. In the carrageenan model of inflammatory pain, ADX-246 increased tolerance to mechanical and thermal pain, and decreased joint swelling. Consistent with previously released data from ADX-629 in a model of alcoholic hepatitis, ADX-246 reduced levels of fibrosis and fat in liver.
“The new data released today support the expansion of our novel RASP platform into clinical indications that may include fat-mass-targeted weight loss and inflammatory pain, highlighting the breadth of potential product candidate opportunities afforded by modulating RASP levels,” stated Todd C. Brady, M.D., Ph.D., President and CEO of Aldeyra.
Based on recent discussions with the U.S. Food and Drug Administration (the FDA), Aldeyra intends to initiate a potentially pivotal Phase 2/3 clinical trial of investigational product candidate ADX-2191 (methotrexate injection, USP) in patients with retinitis pigmentosa due to rhodopsin misfolding mutations. The potential activity of ADX-2191 in retinitis pigmentosa is supported by results from a Phase 2 clinical trial, announced in 2023, which demonstrated improvements from baseline in retinal sensitivity following treatment. An overview of the unmet medical need in retinitis pigmentosa will be provided by Ramiro Maldonado, M.D., Principal Investigator of the Phase 2 clinical trial and Assistant Professor of Ophthalmology, Vitreoretinal Diseases, and Surgery at Duke University.
“Due to loss of vision and dramatic impact on quality of life, retinitis pigmentosa remains a highly significant unmet medical need in retinal disease,” stated Dr. Maldonado. “Even with the advent of gene therapy, cell therapy, and other new approaches not yet approved by the FDA for treatment, a safe and effective drug that could slow the progression of retinitis pigmentosa is in critical demand.”
Research & Development Day Webcast Information
Aldeyra’s Research & Development Day will take place from 9:00 am to 1:00 pm EDT today, Thursday, April 25, 2024, in New York City. A live audio webcast and slide presentation will be accessible from the “Investors & Media” section of the Aldeyra website at https://ir.aldeyra.com/ for 90 days following the event.
About Aldeyra
Aldeyra Therapeutics is a biotechnology company devoted to discovering innovative therapies designed to treat immune-mediated and metabolic diseases. Our approach is to develop pharmaceuticals that modulate protein systems, instead of directly inhibiting or activating single protein targets, with the goal of optimizing multiple pathways at once while minimizing toxicity. Our product candidates include RASP (reactive aldehyde species) modulators ADX-629, ADX-246, ADX-248, and chemically related molecules for the potential treatment of immune-mediated and metabolic diseases. Our late-stage product candidates are reproxalap, a RASP modulator for the potential treatment of dry eye disease and allergic conjunctivitis, and ADX-2191, a novel formulation of intravitreal methotrexate for the potential treatment of retinitis pigmentosa.
Safe Harbor Statement
This release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, including, but not limited to, statements regarding Aldeyra’s future expectations, plans, and prospects, including without limitation statements regarding: the goals, opportunity, and potential for reproxalap, ADX-2191, and other product candidates; the outcome and expected timing and the results of Aldeyra’s planned clinical trials, including planned and ongoing clinical trials for reproxalap and ADX-2191; the outcome and timing of the FDA’s review, acceptance and/or approval of a NDA resubmission for reproxalap and the adequacy of the data included in the original NDA; and the potential NDA resubmission. Aldeyra intends such forward-looking statements to be covered by the safe harbor provisions for forward-looking statements contained in Section 21E of the Securities Exchange Act of 1934 and the Private Securities Litigation Reform Act of 1995. In some cases, you can identify forward-looking statements by terms such as, but not limited to, “may,” “might,” “will,” “objective,” “intend,” “should,” "could," “can,” “would,” “expect,” “believe,” “anticipate,” “project,” “on track,” “scheduled,” “target,” “design,” “estimate,” “predict,” “contemplates,” “likely,” “potential,” “continue,” “ongoing,” “aim,” “plan,” or the negative of these terms, and similar expressions intended to identify forward-looking statements. Such forward-looking statements are based upon current expectations that involve risks, changes in circumstances, assumptions, and uncertainties. Aldeyra is at an early stage of development and may not ever have any products that generate significant revenue. All of Aldeyra's development timelines may be subject to adjustment depending on recruitment rate, regulatory review, preclinical and clinical results, funding, and other factors that could delay the initiation, enrollment, or completion of clinical trials. Important factors that could cause actual results to differ materially from those reflected in Aldeyra's forward-looking statements include, among others, the timing of enrollment, commencement and completion of Aldeyra's clinical trials, the timing and success of preclinical studies and clinical trials conducted by Aldeyra and its development partners; delay in or failure to obtain regulatory approval of Aldeyra's product candidates, including as a result of the FDA not accepting Aldeyra’s regulatory filings, issuing a complete response letter, or requiring additional clinical trials or data prior to review or approval of such filings or in connection with resubmissions of such filings; the ability to maintain regulatory approval of Aldeyra's product candidates, and the labeling for any approved products; the risk that prior results, such as signals of safety, activity, or durability of effect, observed from preclinical or clinical trials, will not be replicated or will not continue in ongoing or future studies or clinical trials involving Aldeyra's product candidates in clinical trials focused on the same or different indications; the scope, progress, expansion, and costs of developing and commercializing Aldeyra's product candidates; uncertainty as to Aldeyra’s ability to commercialize (alone or with others) and obtain reimbursement for Aldeyra's product candidates following regulatory approval, if any; the size and growth of the potential markets and pricing for Aldeyra's product candidates and the ability to serve those markets; Aldeyra's expectations regarding Aldeyra's expenses and future revenue, the timing of future revenue, the sufficiency or use of Aldeyra's cash resources and needs for additional financing; the rate and degree of market acceptance of any of Aldeyra's product candidates; Aldeyra's expectations regarding competition; Aldeyra's anticipated growth strategies; Aldeyra's ability to attract or retain key personnel; Aldeyra’s commercialization, marketing and manufacturing capabilities and strategy; Aldeyra's ability to establish and maintain development partnerships; Aldeyra’s ability to successfully integrate acquisitions into its business; Aldeyra's expectations regarding federal, state, and foreign regulatory requirements; political, economic, legal, social, and health risks, public health measures, and war or other military actions, that may affect Aldeyra’s business or the global economy; regulatory developments in the United States and foreign countries; Aldeyra's ability to obtain and maintain intellectual property protection for its product candidates; the anticipated trends and challenges in Aldeyra's business and the market in which it operates; and other factors that are described in the “Risk Factors” and “Management's Discussion and Analysis of Financial Condition and Results of Operations” sections of Aldeyra's Annual Report on Form 10-K for the year ended December 31, 2023, which is on file with the Securities and Exchange Commission (SEC) and available on the SEC's website at https://www.sec.gov/. Additional factors may be described in those sections of Aldeyra’s Quarterly Report on Form 10-Q for the quarter ended March 31, 2024, expected to be filed with the SEC in the second quarter of 2024, and Aldeyra’s other filings with the SEC.
In addition to the risks described above and in Aldeyra's other filings with the SEC, other unknown or unpredictable factors also could affect Aldeyra's results. No forward-looking statements can be guaranteed and actual results may differ materially from such statements. The information in this release is provided only as of the date of this release, and Aldeyra undertakes no obligation to update any forward-looking statements contained in this release on account of new information, future events, or otherwise, except as required by law.
| Last Trade: | US$4.76 |
| Daily Change: | -0.18 -3.64 |
| Daily Volume: | 591,501 |
| Market Cap: | US$282.840M |
November 18, 2024 October 31, 2024 October 03, 2024 August 08, 2024 | |

ClearPoint Neuro is a global therapy-enabling platform company providing stereotactic navigation and delivery to the brain. Applications of our ClearPoint Neuro Navigation System include electrode lead placement, placement of catheters, and biopsy. The platform has FDA clearance and is...
CLICK TO LEARN MORE
Astria Therapeutics is a biopharmaceutical company, and our mission is to bring life-changing therapies to patients and families affected by rare and niche allergic and immunological diseases. Our lead program, STAR-0215, is a monoclonal antibody inhibitor of plasma kallikrein in clinical development...
CLICK TO LEARN MOREEnd of content
No more pages to load
COPYRIGHT ©2023 HEALTH STOCKS HUB