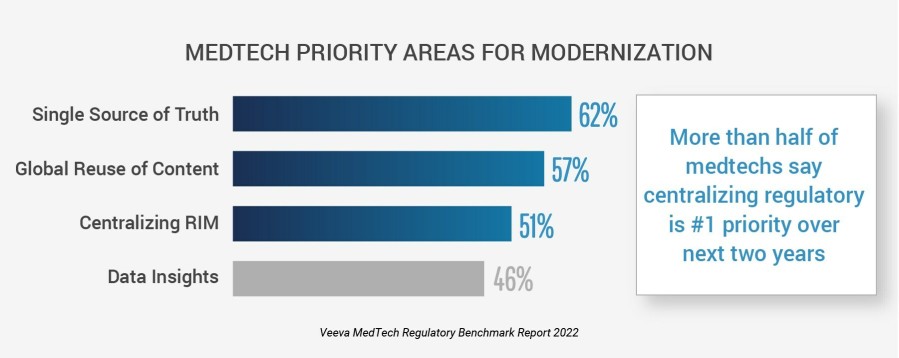
PLEASANTON, Calif., Feb. 28, 2023 /PRNewswire/ -- New research shows a majority of medtech organizations are taking action to advance regulatory affairs, according to the second annual Veeva MedTech Regulatory Benchmark Report. More than half of medtech companies say establishing a single source for regulatory information (62%) and implementing a global and centralized regulatory information management (RIM) system (51%) will be the industry's top focus over the next two years. With the move to modernize RIM underway, medtech leaders are bringing together data, content, and systems for improved insights and time to market.
Data reveals an increased focus on digital systems to harmonize operations globally, with two out of every five companies already selecting digital regulatory technology. This shift toward digital RIM systems highlights the need for greater transparency and collaboration across regional teams to meet evolving regulations and change management requirements.
The report reveals positive changes to regulatory affairs and opportunities for improvement, including:
"With new regulations and increasing supply chain complexity, medtech companies are evaluating paths to more seamless processes so high-quality data can be shared across teams," said Seth Goldenberg, vice president, Veeva MedTech. "This research shows the medtech industry is making progress by advancing regulatory operations for better global visibility, data accuracy, and compliance."
The Veeva MedTech Regulatory Benchmark Report examines the medical device and diagnostic industry's progress toward modernizing regulatory operations. Survey respondents include regulatory affairs professionals from more than 100 medtech organizations around the globe, ranging from enterprise to midsize businesses. See the full annual study which explores how medtech companies manage global compliance and visibility, speed to market, post-market compliance, and modernization.
About Veeva Systems
Veeva (NYSE: VEEV) is the global leader in cloud software for the life sciences industry. Committed to innovation, product excellence, and customer success, Veeva serves more than 1,000 customers, ranging from the world's largest pharmaceutical companies to emerging biotechs. As a Public Benefit Corporation, Veeva is committed to balancing the interests of all stakeholders, including customers, employees, shareholders, and the industries it serves. For more information, visit veeva.com.
Contact:
Deivis Mercado
Veeva Systems
925-226-8821
This email address is being protected from spambots. You need JavaScript enabled to view it.

| Last Trade: | US$213.78 |
| Daily Change: | 3.15 1.50 |
| Daily Volume: | 239,988 |
| Market Cap: | US$34.620B |
November 20, 2024 November 19, 2024 October 24, 2024 September 05, 2024 September 05, 2024 | |

Recursion Pharmaceuticals is a clinical stage TechBio company leading the space by decoding biology to industrialize drug discovery. Enabling its mission is the Recursion OS, a platform built across diverse technologies that continuously expands one of the world’s largest....
CLICK TO LEARN MORE
Astria Therapeutics is a biopharmaceutical company, and our mission is to bring life-changing therapies to patients and families affected by rare and niche allergic and immunological diseases. Our lead program, STAR-0215, is a monoclonal antibody inhibitor of plasma kallikrein in clinical development...
CLICK TO LEARN MOREEnd of content
No more pages to load
COPYRIGHT ©2023 HEALTH STOCKS HUB