LE PONT-DE-CLAIX, France, Sept. 18, 2024 /PRNewswire/ -- BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, announced the commercial release of the BD Neopak™ XtraFlow™ Glass Prefillable Syringe and the latest capacity expansion of the BD Neopak™ Glass Prefillable Syringe platform to serve the growing market for biologic therapies.
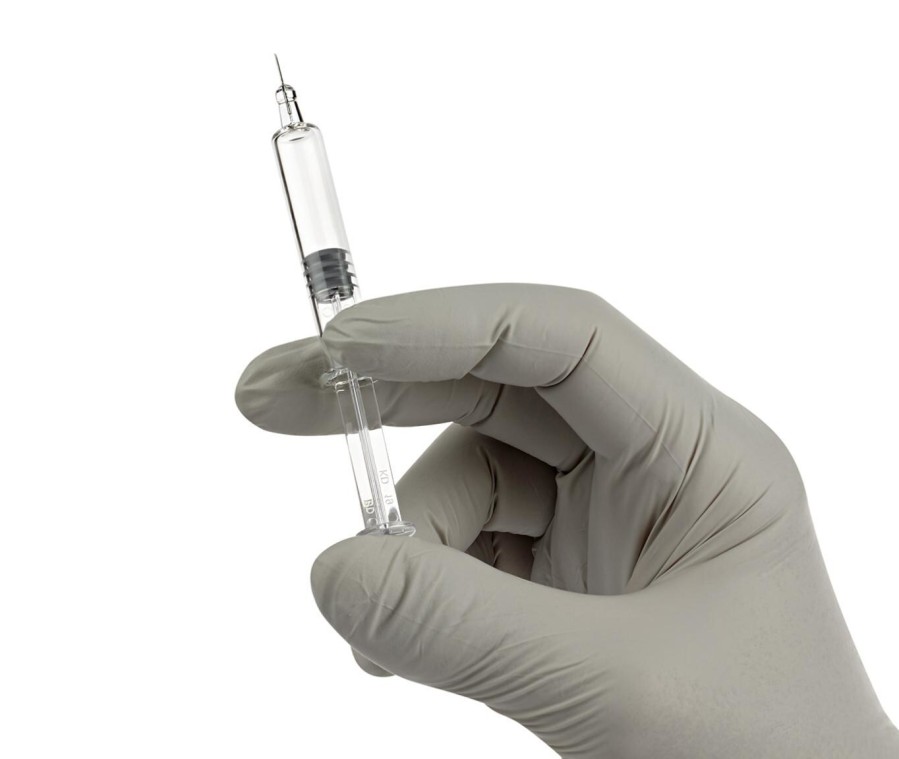
The BD Neopak™ Glass Prefillable Syringe platform is designed to address key development needs for biologic drugs and our customers have received approval to utilize this platform for more than 24 indications, including Crohn's disease, atopic dermatitis, cardiovascular disease and various rare diseases.
BD has expanded usability of this platform with the commercial release of the BD Neopak™ XtraFlow™ Glass Prefillable Syringe. This solution features an 8-millimeter needle length and thinner wall cannula to optimize subcutaneous delivery of higher viscosity drug profiles by reducing the injection force and time required for a fixed solution viscosity1,2*. This improvement allows pharmaceutical developers to break design barriers, enhancing flow and usability beyond today's limits versus a standard half-inch needle3*.
"BD is seeing significant growth in the biologics segment, driven by novel drug formulations across indications such as metabolic disorders and oncology," said Patrick Jeukenne, president of BD Pharmaceutical Systems. "With over 30 drugs approved across 12 markets and many more in development with the BD Neopak™ Glass Prefillable Syringe platform, flexibility is crucial. As a global leader in prefillable drug-delivery systems, BD aims to evolve as rapidly as the global biologics pipeline to enable supply and delivery of these drugs for patients worldwide."
In addition to the usability enhancements, our manufacturing site in Le Pont-de-Claix, France has successfully integrated a high-volume manufacturing line for the BD Neopak™ Glass Prefillable Syringe platform, increasing the production capacity of a single line by sevenfold4. This achievement marks a significant milestone for BD, enabling unprecedented large-scale production of advanced BD glass syringes.
The launch of our BD Neopak™ XtraFlow™ Glass Prefillable Syringe allows developers to take advantage of a single platform approach while dismantling existing design barriers such as drug viscosity. The successful addition of the high-volume line at the Pont-de-Claix site allows BD to be more resilient to supply constraints and further demonstrates our commitment to continuously evolve to meet the needs of our pharmaceutical partners and patients worldwide. These expansions add to BD's leadership in offering a broad range of injection solutions to enable delivery of treatments such as GLP-1s, antibodies, and other next generation treatments for chronic disease.
About BD
BD is one of the largest global medical technology companies in the world and is advancing the world of health by improving medical discovery, diagnostics and the delivery of care. The company supports the heroes on the frontlines of health care by developing innovative technology, services and solutions that help advance both clinical therapy for patients and clinical process for health care providers. BD and its more than 70,000 employees have a passion and commitment to help enhance the safety and efficiency of clinicians' care delivery process, enable laboratory scientists to accurately detect disease and advance researchers' capabilities to develop the next generation of diagnostics and therapeutics. BD has a presence in virtually every country and partners with organizations around the world to address some of the most challenging global health issues. By working in close collaboration with customers, BD can help enhance outcomes, lower costs, increase efficiencies, improve safety and expand access to health care. For more information on BD, please visit bd.com or connect with us on LinkedIn at www.linkedin.com/company/bd1/, X (formerly Twitter) @BDandCo or Instagram @becton_dickinson.
Forward-Looking Statements
This press release contains certain forward-looking statements regarding the BD Neopak™ Glass Prefillable Syringe platform. Forward-looking statements involve risks and uncertainties that could cause actual results to differ materially from those expressed or implied in such statements, including, without limitation, challenges inherent in product development, competitive factors, including the development of new technologies or products by other companies, changes in healthcare and drug delivery, the impact of governmental regulation, future market conditions, manufacturing difficulties or delays, or other factors listed in BD's 2023 Annual Report on Form 10-K and other filings with the SEC. BD expressly disclaims any undertaking to update any forward-looking statements set forth herein to reflect events or circumstances after the date hereof, except as required by applicable laws or regulations.
Contacts: | |
Media: | Investors: |
Fallon McLoughlin | Adam Reiffe |
Director, Public Relations | Sr. Director, Investor Relations |
201.258.0361 | 201.847.6927 |
This email address is being protected from spambots. You need JavaScript enabled to view it. | This email address is being protected from spambots. You need JavaScript enabled to view it. |
1. Injection time and ejection force calculation [internal study], Le Pont-de-Claix, France; Becton, Dickinson and Company, 2021
2. BD NeopakTM XtraFlowTM 2.25 mL prototype evaluation [internal study] , Le Pont-de-Claix, France; Becton, Dickinson and Company, 2020
3. Pager et al. (2020), "User experience for manual injection of 2 mL viscous solutions is enhanced by a new prefillable syringe with staked 8 mm ultra-thin wall needle", Expert Opinion on Drug Delivery, https://doi.org/10.1080/17425247.2020.1796630.
4. BD data on file
*When compared to 12.7 mm special thin wall needle

| Last Trade: | US$225.15 |
| Daily Change: | -2.02 -0.89 |
| Daily Volume: | 1,682,996 |
| Market Cap: | US$65.080B |
November 07, 2024 October 30, 2024 October 29, 2024 October 14, 2024 September 26, 2024 | |

Astria Therapeutics is a biopharmaceutical company, and our mission is to bring life-changing therapies to patients and families affected by rare and niche allergic and immunological diseases. Our lead program, STAR-0215, is a monoclonal antibody inhibitor of plasma kallikrein in clinical development...
CLICK TO LEARN MORE
C4 Therapeutics is pioneering a new class of small-molecule drugs that selectively destroy disease-causing proteins via degradation using the innate machinery of the cell. This targeted protein degradation approach offers advantages over traditional drugs, including the potential to treat a wider range of diseases...
CLICK TO LEARN MOREEnd of content
No more pages to load
COPYRIGHT ©2023 HEALTH STOCKS HUB