TOKYO and EMERYVILLE, Calif., July 10, 2023 /PRNewswire/ -- Astellas Pharma Inc. (TSE: 4503) (President and CEO: Naoki Okamura, "Astellas") and 4D Molecular Therapeutics, Inc. (NASDAQ: FDMT) (CEO: David Kirn, MD, "4DMT") today announced a license agreement under which Astellas gains rights to utilize the intravitreal retinotropic R100* vector invented by 4DMT for one genetic target implicated in rare monogenic ophthalmic disease(s), with options to add up to two additional targets implicated in rare monogenic ophthalmic diseases after paying additional option exercise fees.
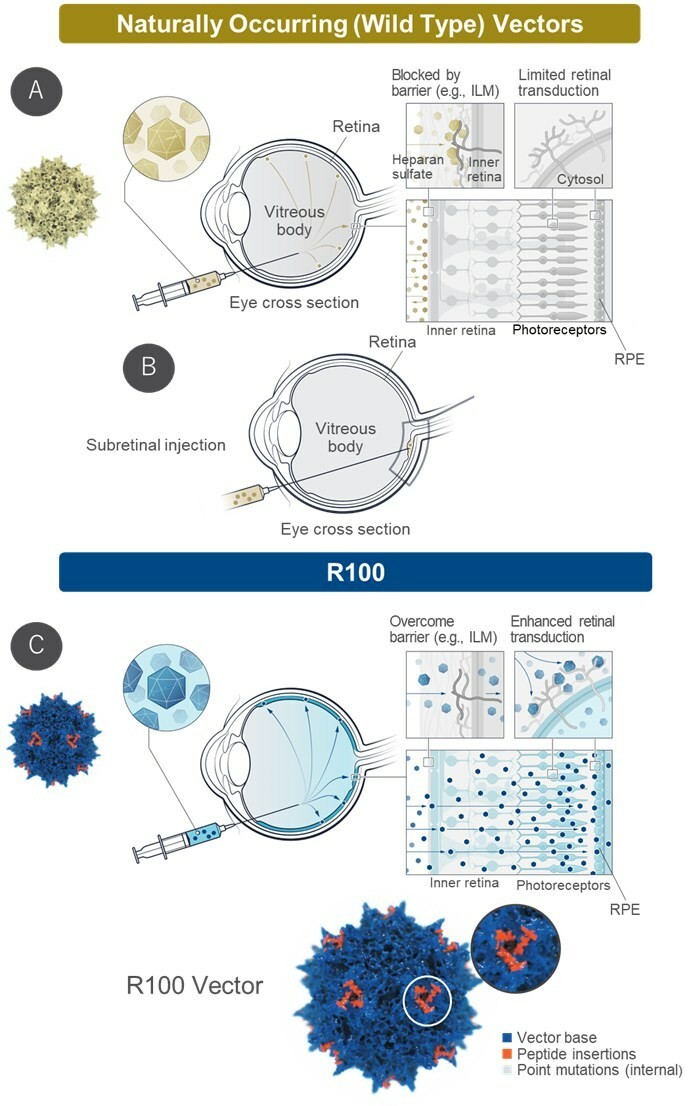
R100 is an adeno-associated virus (AAV) vector invented by 4DMT for intravitreal delivery. It has the ability to penetrate the internal limiting membrane barrier and to efficiently transduce the entire retina, resulting in robust transgene expression within retinal cells. All three 4DMT clinical-stage ophthalmic product candidates utilize the R100 vector, including 4D-150 for wet age-related macular degeneration and diabetic macular edema.
Under the terms of the agreement, 4DMT will provide its proprietary R100 vector technology to Astellas to deliver Astellas' unique genetic payloads for the treatment of rare monogenic diseases. Astellas will conduct all subsequent research, development, manufacturing, and commercialization activities. 4DMT will receive US$20 million upfront, and potential future option fees and milestones of up to US$942.5 million including potential near-term development milestones of US$15 million for the initial target. In addition, 4DMT is entitled to receive mid-single digit to double-digit, sub-teen royalties on net sales of all licensed products.
"This collaboration with Astellas, a leader in AAV gene therapy, continues to validate R100 for routine intravitreal low dose delivery of genetic payloads for the treatment of retinal diseases," said David Kirn, M.D., Co-Founder and Chief Executive Officer of 4DMT. "With over 70 patients dosed to-date with R100-based product candidates in wet age-related macular degeneration and rare ophthalmic diseases, this collaboration also demonstrates the modularity of the Therapeutic Vector Evolution platform resulting in efficient design and development of new intravitreal products. 4DMT retains rights to large market non-hereditary ophthalmic diseases."
Adam Pearson, Chief Strategy Officer (CStO) at Astellas said, "At Astellas, we have a strong commitment to developing novel treatments for ophthalmic diseases, and have positioned Blindness & Regeneration as one of the Primary Focuses of our R&D strategy. Staying at the forefront of gene therapy technology is a key part of our strategy. We believe that this collaboration will bring synergies between the two companies' cutting-edge research, and will ultimately lead to the development of new therapeutics for patients with ophthalmic diseases at high risk of blindness."
About Astellas
Astellas Pharma Inc. is a pharmaceutical company conducting business in more than 70 countries around the world. We are promoting the Focus Area Approach that is designed to identify opportunities for the continuous creation of new drugs to address diseases with high unmet medical needs by focusing on Biology and Modality. Furthermore, we are also looking beyond our foundational Rx focus to create Rx+® healthcare solutions that combine our expertise and knowledge with cutting-edge technology in different fields of external partners. Through these efforts, Astellas stands on the forefront of healthcare change to turn innovative science into VALUE for patients. For more information, please visit our website at https://www.astellas.com/en.
About 4DMT
4DMT is a clinical-stage biotherapeutics company harnessing the power of directed evolution for genetic medicines targeting large market diseases. 4DMT seeks to unlock the full potential of genetic medicines using its proprietary invention platform, Therapeutic Vector Evolution, which combines the power of the Nobel Prize-winning technology, directed evolution, with approximately one billion synthetic AAV capsid-derived sequences to invent customized and evolved vectors for use in our product candidates. All of our vectors are proprietary to 4DMT and were invented at 4DMT, including the vectors utilized in our clinical-stage and preclinical pipeline product candidates: R100, A101, and C102. The Company is initially focused on five clinical-stage product candidates in three therapeutic areas for both rare and large market diseases: ophthalmology, pulmonology, and cardiology. The 4DMT customized and evolved vectors were invented with the goal of being delivered at relatively low doses through clinically routine, well-tolerated, and minimally invasive routes of administration, transducing diseased cells in target tissues efficiently, having reduced immunogenicity and, where relevant, having resistance to pre-existing antibodies. 4DMT is currently advancing five product candidates in clinical development: 4D-150 for wet AMD and DME, 4D-710 for cystic fibrosis lung disease, 4D-310 for Fabry disease cardiomyopathy, 4D-125 for XLRP, and 4D-110 for choroideremia. The 4D preclinical product candidates in development are: 4D-175 for geographic atrophy and 4D-725 for AATLD.
4D-150, 4D-710, 4D-310, 4D-125, and 4D-110 are our product candidates in clinical development and have not yet been approved for marketing by the US FDA or any other regulatory authority. No representation is made as to the safety or effectiveness of 4D-150, 4D-710, 4D-310, 4D-125, or 4D-110 for the therapeutic uses for which they are being studied.
4D Molecular Therapeutics™, 4DMT™, Therapeutic Vector Evolution™, and the 4DMT logo are trademarks of 4DMT.
Cautionary Notes (Astellas)
In this press release, statements made with respect to current plans, estimates, strategies and beliefs and other statements that are not historical facts are forward-looking statements about the future performance of Astellas. These statements are based on management's current assumptions and beliefs in light of the information currently available to it and involve known and unknown risks and uncertainties. A number of factors could cause actual results to differ materially from those discussed in the forward-looking statements. Such factors include, but are not limited to: (i) changes in general economic conditions and in laws and regulations, relating to pharmaceutical markets, (ii) currency exchange rate fluctuations, (iii) delays in new product launches, (iv) the inability of Astellas to market existing and new products effectively, (v) the inability of Astellas to continue to effectively research and develop products accepted by customers in highly competitive markets, and (vi) infringements of Astellas' intellectual property rights by third parties.
Information about pharmaceutical products (including products currently in development) which is included in this press release is not intended to constitute an advertisement or medical advice.
4D Molecular Therapeutics' Forward Looking Statements:
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including, without limitation, implied and express statements regarding the therapeutic potential, abilities, safety and efficacy of 4D Molecular Therapeutics' proprietary intravitreal R100 vector, including in connection with Astellas' use thereof pursuant to the License Agreement, the potential benefits or applications of 4D Molecular Therapeutics' Vector Evolution platform, including any other vectors developed through the Vector Evolution platform, and the amount of potential milestone and option payments payable under the License Agreement. The words "may," "might," "will," "could," "would," "should," "expect," "plan," "anticipate," "intend," "believe," "expect," "estimate," "seek," "predict," "future," "project," "potential," "continue," "target" and similar words or expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. Any forward looking statements in this press release are based on management's current expectations and beliefs and are subject to a number of risks, uncertainties and important factors that may cause actual events or results to differ materially from those expressed or implied by any forward-looking statements contained in this press release, including risks and uncertainties that are described in greater detail in the section entitled "Risk Factors" in 4D Molecular Therapeutics' most recent Annual Report on Form 10-K and Quarterly Report on Form 10-Q, as well as any subsequent filings with the Securities and Exchange Commission. In addition, any forward-looking statements represent 4D Molecular Therapeutics' views only as of today and should not be relied upon as representing its views as of any subsequent date. 4D Molecular Therapeutics explicitly disclaims any obligation to update any forward-looking statements. No representations or warranties (expressed or implied) are made about the accuracy of any such forward looking statements.

| Last Trade: | US$7.50 |
| Daily Change: | -0.12 -1.57 |
| Daily Volume: | 685,897 |
| Market Cap: | US$389.780M |
November 13, 2024 September 23, 2024 September 18, 2024 August 27, 2024 | |

Compass Therapeutics is a clinical-stage, oncology-focused biopharmaceutical company developing proprietary antibody-based therapeutics to treat multiple human diseases. The company's scientific focus is on the relationship between angiogenesis, the immune system, and tumor growth...
CLICK TO LEARN MORE
Astria Therapeutics is a biopharmaceutical company, and our mission is to bring life-changing therapies to patients and families affected by rare and niche allergic and immunological diseases. Our lead program, STAR-0215, is a monoclonal antibody inhibitor of plasma kallikrein in clinical development...
CLICK TO LEARN MOREEnd of content
No more pages to load
COPYRIGHT ©2023 HEALTH STOCKS HUB